 
He was a good lecturer, encouraged his students, and devoted considerable attention to the wider problems of science teaching at university and secondary levels. Cathode rays move opposite to anode rays. Later it was found that anode rays and Cathode rays both exist at the same time. He built his cathode ray tube with a metal cylinder on the other end. Even though he was clumsy with his hands, he had a genius for designing apparatus and diagnosing its problems. JJ Thomson did an experiment with discharge tube using hydrogen gas. Thompson, conducted his first cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from the latent charge. Image: Draw a fine-beam tube with annotations/labels. In 1884 he was named to the prestigious Cavendish Professorship of Experimental Physics at Cambridge, although he had personally done very little experimental work. What four things did JJ Thomson demonstrate about cathode rays 1. He was then recommended to Trinity College, Cambridge, where he became a mathematical physicist. Thomson gave a lecture detailing his and others’ experiments with the energetic beams inside cathode-ray tubes. Instead young Thomson attended Owens College, Manchester, which had an excellent science faculty. Exactly 125 years ago, the British physicist J.J. His father intended him to be an engineer, which in those days required an apprenticeship, but his family could not raise the necessary fee. Learn all about the discovery, the importance of the discovery, and JJ Thomson in this tutorial article. Ironically, Thomson-great scientist and physics mentor-became a physicist by default. JJ Thomson made the discovery using the cathode ray tube. His assistant, Francis Aston, developed Thomson’s instrument further and with the improved version was able to discover isotopes-atoms of the same element with different atomic weights-in a large number of nonradioactive elements. Here his techniques led to the development of the mass spectrograph. Thomson’s last important experimental program focused on determining the nature of positively charged particles. His efforts to estimate the number of electrons in an atom from measurements of the scattering of light, X, beta, and gamma rays initiated the research trajectory along which his student Ernest Rutherford moved. He used them in his experiments with cathode ray tubes in order to determine what cathode rays were composed of. In 1904 Thomson suggested a model of the atom as a sphere of positive matter in which electrons are positioned by electrostatic forces. While working in Thomson’s laboratory shortly after Thomson discovered the electron, Rutherford showed that compounds of uranium and other elements emitted at least two distinct types of radiation.Structure of the Atom and Mass Spectrography This 1922 photo was taken in the dark in the Curie laboratory.īuilding on the Curies’ work, the British physicist Ernest Rutherford (1871–1937) performed decisive experiments that led to the modern view of the structure of the atom. 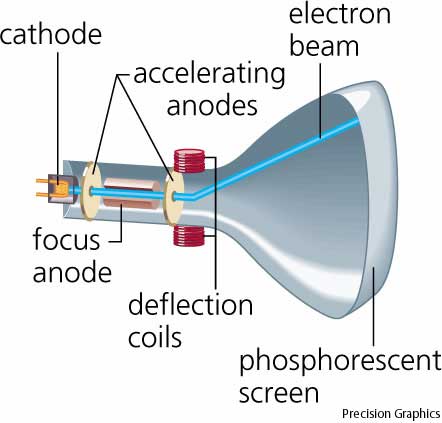
The apparatus of his experiment is called the cathode-ray tube (CRT). These particles later were named electrons. He shot tiny things called alpha particles at a thin sheet of gold foil. In 1897, he showed that cathode rays were composed of very small negatively charged particles. He was well-known for the discovery of the electron. \): Radium bromide illuminated by its own radioactive glow. Physical Chemistry Sir Joseph John Thomson was a British physicist and Nobel Laureate. Cathode Ray Tube - The Cathode Ray Experiment by J.J.Thomson helped to discover electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
